
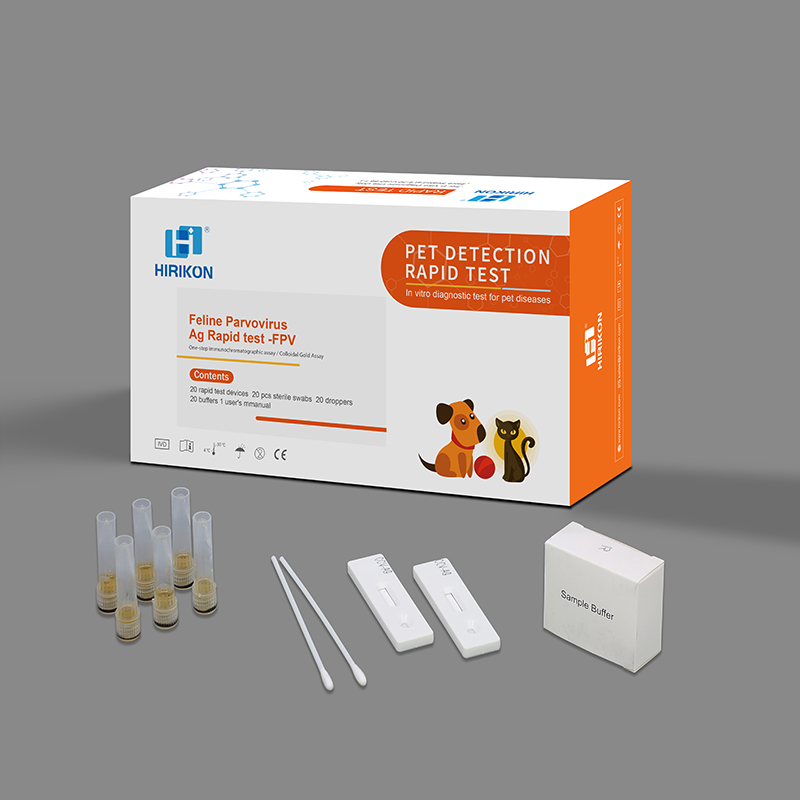
Feline ParvovirusAg Rapid test -FPV
Place of Origin:China
Brand Name:HIRIKON
Properties:Diagnosis & Injection
Purpose:Detection of Feline panleukopenia virus antigen
Principle:Immunochromatographic assay
Specimen:feline feces
Sensitivity:97%
Specificity:98.5%
Materials providedTest card, diluents tube
Accessories:disposable swab and dropper
Test time:10-15 mins
Feline Panleukopenia virus Antigen test kit fpv Ag rapid Diagnostic test
INTENDED USE
Feline Parvovirus Ag Test is designed for the qualitative detection of parvovirus antigen (FPV Ag) in animal’s feces or serum according to sandwich lateral flow immunochromatographic principle.
Assay Time: 5-10 min
Sample: Feces
PRINCIPLE OF THE ASSAY
Feline Parvovirus Ag Test is a sandwich lateral flow immunochromatographic assay. There is a testing window on the test
device which contains invisible T (test) zone and C (control) zone. When sample is applied into the sample hole on the device, the
liquid will laterally flow on the surface of the test strip. If there is enough parvovirus antigen in the sample, a visible T band
will appear. The C band should always appear after a sample is applied, indicating a valid result. By this means, the device can
accurately indicate the presence of parvovirus antigen in the sample.
TEST PROCEDURE
1st Step
– Collect animal’s fresh feces with a swab stick.
2nd step
– Insert the wet swab into the provided tube with assay Agitate it to assure good sample extraction.
3rd Step
– If using serum sample, please do a dilution of 1:3 with the provided assay buffer.
4th Step
– Gradually drip 2-3 drops of sample extraction into the sample hole.
5th Step
Interpret the result in 5-10 minutes. Result after 10 minutes is considered as invalid.
STORAGE
The kit can be stored at room temperature (2-30°C). The test kit is stable through the expiration date (18 months) marked on the
foil pouch. DO NOT FREEZE. Do not store the test kit in direct sunlight.
PRECAUTIONS
– For best results, please strictly adhere to these instructions.
– All reagents must be placed at room temperature before running the assay.
– Do not remove test cassette from its pouch until immediately before use.
– Do not reuse the test kit.
– Do not use the test beyond its expiration date marked on the foil pouch.
– The components in this kit have been quality control tested as standard batch unit. Do not mix components from different lot
numbers.







Reviews
There are no reviews yet.