
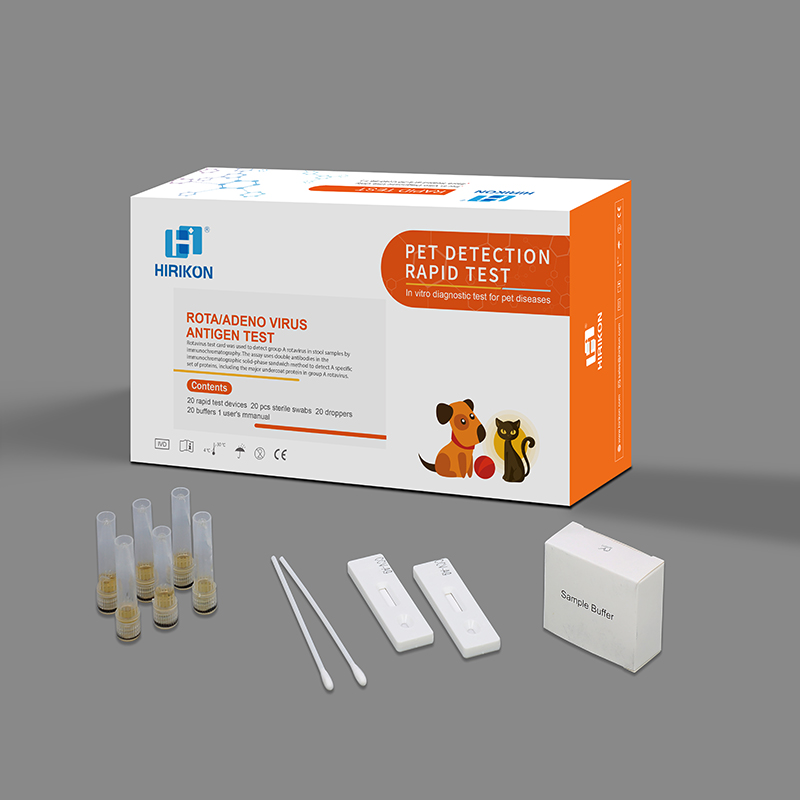
ROTA/ADENO VIRUS ANTIGEN TEST
Place of Origin:China
Brand Name:HIRIKON
Model Number:ROTA/ADENO
Power Source:Manual
Warranty:2 Years
After-sale Service:Online technical support
Material:Plastic, Medical Grade ABS Plastic
Shelf Life:2 years
Quality Certification:ce
Instrument classification:Class II
Product name:rota/adeno
Certificate:CE
Function:Early Detection
Format:cassette
Package:Single Pouch + Carton
Accuracy:99%
Display:screening
Method:Colloidal Gold Rapid Test
High quality Adeno/rota Antigen Feces 2 in 1 Muti-Panel Combo Cassette
The Adenovirus is the second most common cause of viral gastro-enteritis in children (10-15%). This virus may also cause respiratory diseases and, depending on the serotype, also diarrhea, conjunctivitis, cystitis, etc. At least 47 serotypes of adenovirus have been described, all sharing a common hexon antigen. Serotypes 40 and 41 are the ones associated with gastro-enteritis, whose main symptom is diarrhoea that may last between 9 and 12 days associated with temperature and vomits.
SUMMARY AND PRINCIPLE OF THE TEST
The test uses new homogenous immunochromatographic system with gold particules. It is a ready to use test which only needs a faecal sample dilution with the supplied ready to use dilution buffer. Specificity is ensured by using a monoclonal antibody conjugated with gold particules and directed against specific human genus-specific Rotavirus antigen or Adenovirus antigens. The immunochromatographic stick is coated with a monoclonal immunoreagent specific for genus-specific Adenovirus hexon antigens or rotavirus VP6 antigen. Liquid sample and gold conjugate both migrate by capillarity and reach the first specific anti-Adenovirus monoclonal reagent. If Adenovirus is present in the sample, it is blocked and immunoreactions appear as a red-pink line. As sample still migrates, it reaches the second non specific anti-mouse IgG which gives rise to a second red-pink line. This rear line indicates that the chromatography has been developed without hindrance. It appears also with negative samples.
PRECAUTION FOR USERS
1. For in-vitro diagnostic use only.
2. Handle all specimens as if they contain infectious agents.When the assay procedure is completed, dispose ofspecimens carefully after autoclaving for at least one hour.Alternatively, treat with a 0.5 or 1% solution of sodiumhypochlorite for one hour before disposal.
3. Wear protective clothing (laboratory coats and disposablegloves) when assaying samples.
ASSAY PROCEDURES
1. Allow all reagents to reach room temperature before use.
2. Place 0.5-1ml of extraction buffer in a properly marked testing tube.
3. Add a sample portion of approximately 5-6 mm size (25-100mg), with a swab, a wooden applicator or a bacteriology loop. Press the applicator to the tube and rotating it at the same time. For liquid or semi-solid stools add 100 micro liters of stool using an appropriate pipette.
4. Shake vigorously in order to resuspend it into the buffer. If needed Vortex for 15 seconds.
5. Dip the reaction strip in the test tube with the arrow pointing to the bottom.
6. Incubate the test at room temperature and read the test in 5-15 minutes.
Note: Results after 15 minutes may not be accurate.
INTERPRETATION OF RESULTS
Negative: One pink line appears in control line, showing the test has been carried out correctly. There will be no line in test region
Positive: In addition to a pink colored control line, for adenovirus positive, an upper test line will appear, for rotavirus positive; a lower test line will appear.
Invalid: A total absence of color in both regions is an indication of procedure error and/or that the test reagent has deteriorated. The test should be repeated using a new cassette.







Reviews
There are no reviews yet.